Category Archives: Allergy
FaRMI
I will add now a special collection of farming studies here as many of them are just candidates for the Ig nobel prize.
The most recent study introduces FaRMI, a “bacterial relative abundance farm home microbiota index”, probably introduced as the authors couldn’t find anything else. It reminds me very much to the polygenic risk score that rescues your study if you could not find the gene.
Asthma prevalence has increased in epidemic proportions with urbanization
Already the first sentence is wrong if we look at the following plot where asthma is clearly levelling off.
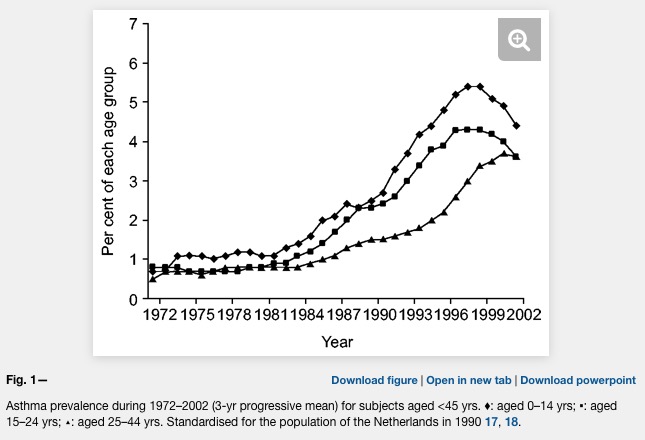
Unfortunately, the difference between farm and non farm children is never explained in the Kirjavainen et al. paper. What is the average distance of a non farm house to a farm house? Are there any joint school or sports activities of children from farms and non farms (allergens travel in the classroom)? And why is there such a strong conclusion in the title?
Farm-like indoor microbiota in non-farm homes protects children from asthma development
A lower risk score is not equivalent to protection.
And did any reviewer ever look at the plots or tables?
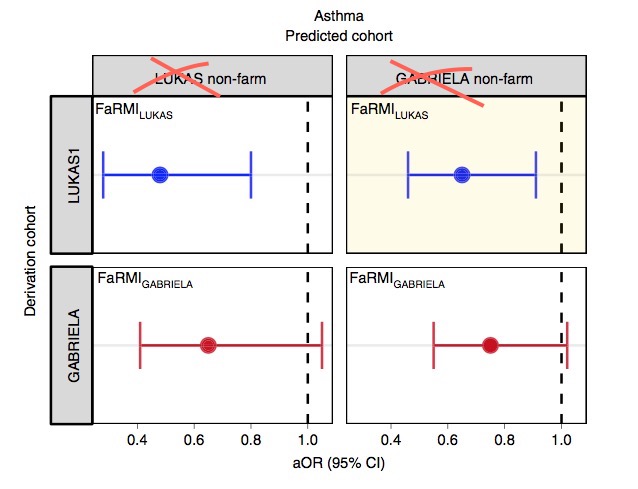
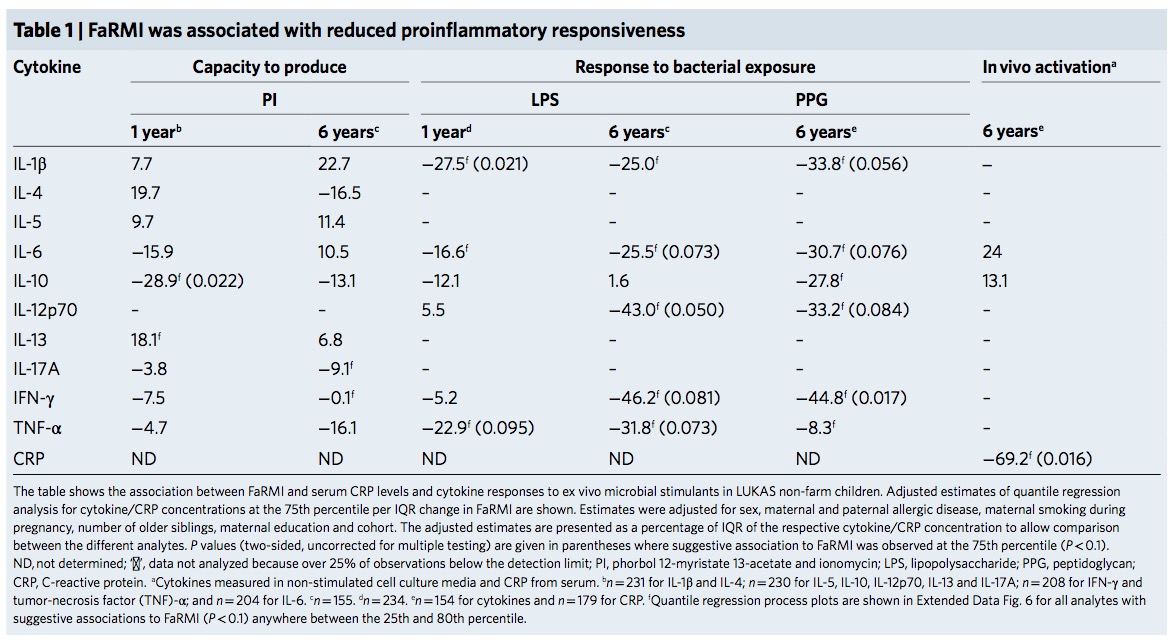
The FaRMI calculation is overly complicated. Probably the calculation can never be replicated by changing software, changing samples, unclear wording, suboptimal dimension reduction and data snooping.
Farm home microbiota-like community composition was modeled in LUKAS1 with logistic regression analysis (PROC LOGISTIC statement, SAS version 9.3). The home location on a farm or non-farm rural environment was the dependent variable and the main components of PCoA axis scores of β-diversity matrices were the predictor variables. Bacterial and fungal microbiota were investigated separately. For both bacteria and fungi, separate models were built using axis scores from PCoA of abundance-unweighted and -weighted β-diversity matrices. The PCoA axes were selected based on the scree plot method including axes above the point at which the variance explained by the additional axes levels off (Supplementary Fig. 3). The models give an estimate of the probability that the sample is from a farm home. The farm home likeness of the microbial composition in the LUKAS2 non-farm homes was then estimated by applying the regression coefficients obtained from the LUKAS1-based models to the corresponding microbial data from LUKAS2 samples.Some analyses were performed in non-farm homes of both LUKAS2 and LUKAS1 to obtain increased sample size and power if results remain comparable as was observed. Due to the discovered association with asthma, the probability that was modeled based on the relative abundance-weighted bacterial/archaeal β-diversity was named FaRMI and was studied further in greater detail.
And isn’t that just an association that may have a rather simple explanation?
As FaRMI is weakly associated with muramic acid concentration in dust, the authors make Gram-positive bacteria responsible for the effect. The rhizosphere of soil is extremly rich of bacteria. The world’s first soil atlas showed hundreds of taxa but never differentiated between water resistant, gram positive and less water resistant gram-negative taxa. Maybe Gram positive Streptococcaceae are ubiquitous and depend on where you draw your samples?
FaRMI is found in non farm / rural children by bacterial/archaeal operational taxonomic units (OTUs) of soil origin which basically confirms my initial assumption: There was the same contamination of soil both in farm and non-farm homes if we look at supplement table 6 where walking indoors with outdoor shoes results in significant higher FaRMI values…Maybe the microbiome hype is already over.
Our results warrant translational studies to confirm the causal relationship through indoor microbial exposure-modifying intervention that may also form a novel strategy for primary asthma prevention.
Good luck with your future studies.
BTW – The scripts at Github are useless references to shell and Python scripts that will never run due to “—” characters. And what about that baby code?
outfolder=getwd()
eigenfile <- paste(outfolder, "/", prefix, "_PCoA_eigenvalues.txt", sep="")
writerow <- paste("Eigenvalue min / max: ", min.eigen, " / ", max.eigen, sep="")
write(writerow, file=eigenfile, append=F)
writerow <- paste("Sum of all eigenvalues: ", round(neg.eigensum, digits=6), sep="")
write(writerow, file=eigenfile, append=T)
writerow <- paste("Sum of all eigenvalues (negatives as 0): ", round(nonneg.eigensum, digits=6), sep="")
write(writerow, file=eigenfile, append=T)
writerow <- "Eigenvalues (pos & neg): "
write(writerow, file=eigenfile, append=T)
writerow <- paste(pcoa$value$Eigenvalues, collapse="\t")
write(writerow, file=eigenfile, append=T)
writerow <- "Percents (Negatives as negatives): "
write(writerow, file=eigenfile, append=T)
writerow <- paste(paste(neg.percent, " %", sep=""), collapse="\t")
write(writerow, file=eigenfile, append=T)
writerow <- "Percents (Negatives as 0): "
write(writerow, file=eigenfile, append=T)
writerow <- paste(paste(nonneg.percent, " %", sep=""), collapse="\t")
write(writerow, file=eigenfile, append=T)
Using R heredoc syntax I can rewrite 20 unreadable by 8 readable lines.
tmp <- 'Eigenvalue min / max: min.eigen / max.eigen
Sum of all eigenvalues: neg.eigensum
Sum of all eigenvalues (negatives as 0): nonneg.eigensum
Eigenvalues (pos & neg): pcoa
Percents (Negatives as negatives): neg.percent %
Percents (Negatives as 0): nonneg.percent %'
for (i in c("min.eigen","max.eigen","neg.eigensum","nonneg.eigensum","pcoa$value$Eigenvalues","neg.percent","nonneg.percent") ) { tmp <- gsub(i,get(i),tmp) }
write(tmp, file=paste(getwd(),out,"/")
And why moving to SAS for a simple logistic regression? Is there anyone else in the academic world who pays $8,700 annually for a basic SAS Windows Analytics package just for that reason?
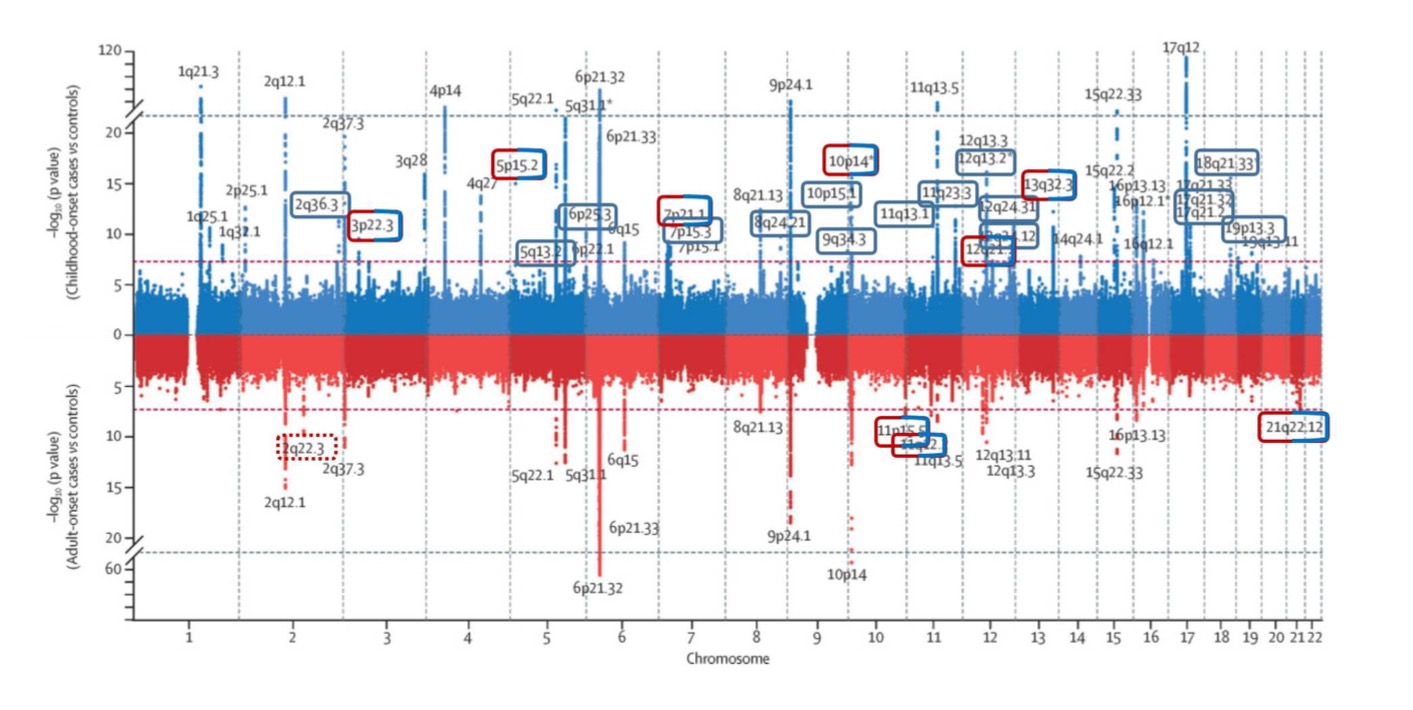
Asthma genetics: Bigger is not better
20 years ago with limited funds we published hits on chromosome 2, 6, 9 and 12.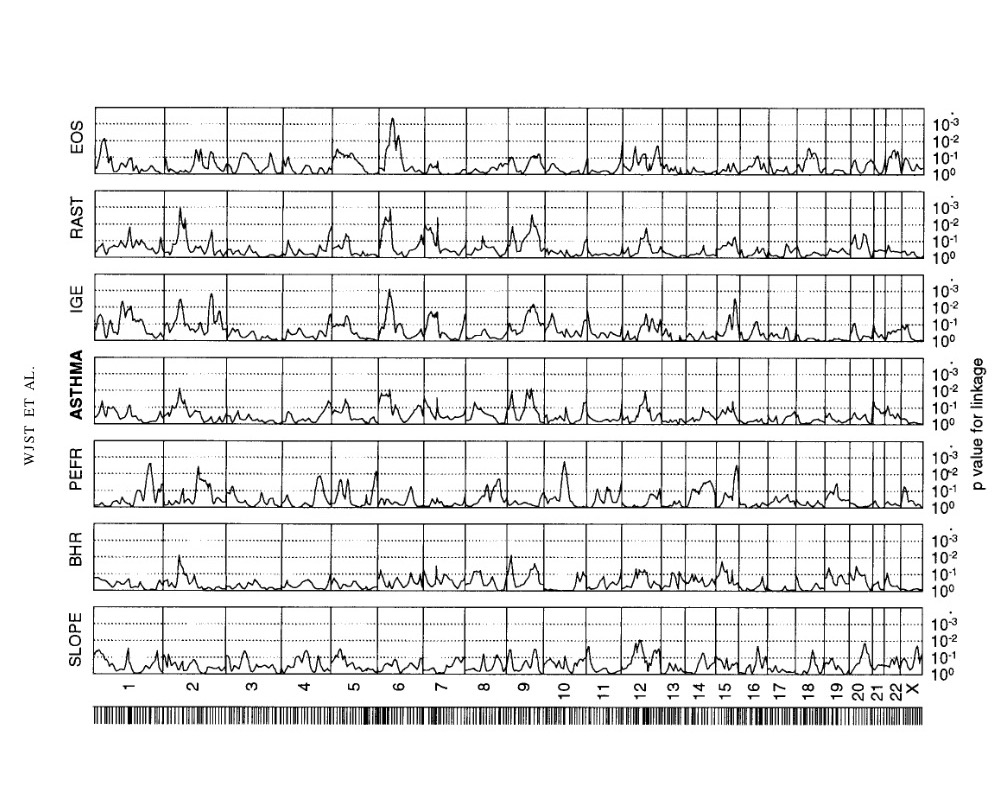
Yesterday I have seen this plot. The lower part is adult asthma: hits on chromosome 2, 6, 9, 10, 12.
We needed N=415, the plot below needed N=500,000 individuals.
Forget about plasma vitamin D measurements
I have written in my recent editorial about the nonsense of plasma vitamin D measurements. A recent case history of a patient with a deleted vitamin D carrier molecule GC now confirms the free-hormone hypothesis. The patient’s plasma 25(OH)D levels was only 0.4% of those in the unaffected sibling.
Despite a lifelong deficiency of vitamin D binding protein, limited sun exposure (for religious reasons), and a diet that was probably lacking sufficient vitamin D, our patient did not have rickets or osteomalacia but rather osteopenia and fragility fractures that occurred in the fifth decade of life.
Another carrier sibling had only two third of the plasma 25(OH)D level compared to the unaffected sibling but showed “no appreciable clinical manifestations”.
So why measure 25(OH)D?
IL33, allergy and helminths: Shot in the leg?
Ever since our NEJM paper in 2010 that showed an IL33/ST2 association there are new studies on IL33.
Grotenboer 2013 did a functional annotation of the gene and it’s receptor in humans while there is no more doubt about the involvement of IL33 in human allergy. Right now IL33 suppression is already used as an experimental screening test for allergic reactivity with ongoing phase II studies of anti-IL33 or anti ST2. Good IL33 reviews can be found for example in frontiers in immunology by Tataori et al. or in nature immunology by Smith.
These reviews do not tell you so much about the regulation while regulation has recently elucidated by Gour et al. who describe a tropomyosin–dectin-1 interaction of the human host. Why is tropomyosin such a frequent target of human IgE?
Muscle protein tropomyosin is an important IgE target in a number of nematode infections; Onchocerca volvulus ; Ascaris lumbricoides; Anisakis simplex; and tropomyosin from the blood fluke Schistosoma mansoni is also a human IgE antigen. Tropomyosin is highly conserved across many invertebrates and is responsible for much of the IgE cross-reactivity between Ascaris and dust-mites.
I haven’t found any good answer to this question. As tropomyosin affects contractility – this seems like “shooting into the leg” of worms whenever they attempt to invade.
Maybe Gour et al. did not know the earlier dissertation from Berlin that already showed a reduced inflammation in the OVA mouse model by administration of recombinant tropomyosin.
The broad cross reactivity to tropomyosin gives rise to the question if helminth tropomyosin could induce allergic reactions to itself and/or tropomyosin of different organisms. Considering the fact that filarial nematodes express tropomyosin on their surface […] and that the continuing turnover of microfilariae confronts the host with relevant amounts of tropomyosin makes this question even more appropriate.
Worms seems to be attacked by anti-worm-surface-tropomyosin IgE whenever the worm tries to invade the epithelium during an acute infection. During invasion extracellular IL33 is cleaved into a shorter form with enhanced activity attracting more immune cells.
During chronic infestation nothing happens as long as the worm does not invade and doesn’t trigger any IL33 alarmin. As there is continuous tropomyosin antigen antigen contact, the host is slowly desensitzed, clearing IgE in favor of IgG4.
Is this also a model that explains allergy? We don’t know the details but maybe this antigen recognition / response system is being disturbed where allergens like Der p1 mimicking a worm infection by tropomyosin can trigger the allergic reaction in particular as Der p1 a cysteine protease also mimicks an invasion signal.
23.12.2019 Addendum
Parasite tropomyosin ist detected in in 55%-62% of patients (cockroach tropomyosin rPer a 7, Ascaris tropomyosin rAsc l 3).
RCT No4 shows vitamin D supplement as allergy risk
I nearly forgot in my recent list of RCTs to mention the new study of Jenni Rosendahl who compared high-dose to low-dose vitamin D supplementation effects.
The Vitamin D Intervention in Infants (VIDI) study was a randomized controlled 24-month trial of daily 400 IU or 1200IU vitamin D supplementation administered to healthy infants that evaluated the effect of vitamin D supplementation on bone health and infections.
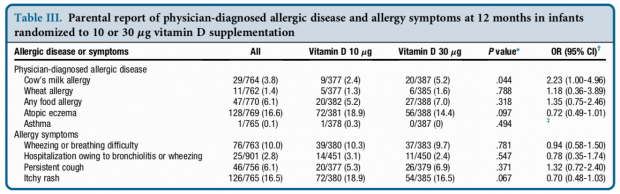
I would say sensitization against one of the most common allergen in this age group happens already in the low dose group while the higher dose leads to a further 2.2fold increased disease risk.
Would be really important to have any unexposed control group here.
Single vitamin D bolus and HLA accessible chromatin
We have already recently seen that gene methylation in newborns can be changed by maternal vitamin D supplementation.
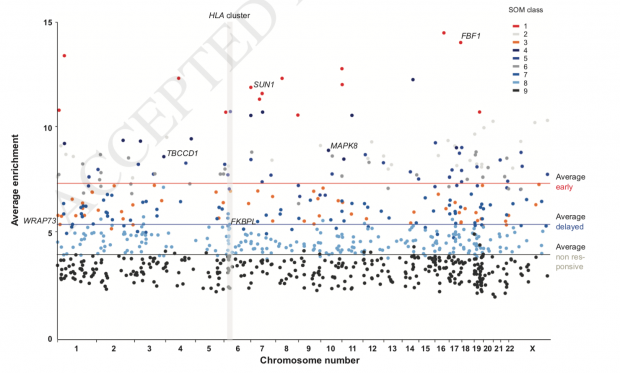
This is now confirmed in a single individual who was exposed to an oral bolus of 2000 μg of vitamin D3. Even within one day, effects could be observed.
Consistently accessible chromatin was detected at 5,205 genomic loci, the 853 most prominent of which a self-organizing map algorithm classified into early, delayed and non-responding genomic regions: 70 loci showed already after one day and 361 sites after two days significant (p < 0.0001) chromatin opening or closing. Interestingly, more than half of these genomic regions overlap with transcription start sites, but the change of chromatin accessibility at these sites has no direct effect on the transcriptome.
Early responses are described for SUN1 (funny in this context :-), FBF1 and WRAP73. Overall the genomic region around the human leukocyte antigen (HLA) cluster in chromosome 6 showed the highest normalized density of accessible chromatin explaining the immunosuppressive effect of sunshine.
Allergy and vitamin D supplements only in domesticated animals
This is an argument that I found only recently in the excellent review by Hellman 2017
Both humans and rodents living under laboratory conditions are generally free from worm infections, which are known to be potent inducers of IgE production. By contrast, most wild animal populations have massive amounts of intestinal worm parasites … To our knowledge, allergies have not been described in wild animals. One potential factor could be a genetic drift due to strong selection for phenotypic characteristics like coat color, long or short noses, running fast, or wanted social behaviors. Such strong selections are seen in the breeding programs for dogs, horses, and cats, but a questionable cause for human allergies. However, it is possible that we constantly need to be selecting against hypersensitivities, which may occur due to minor shift in immune functions caused by spontaneous point mutations. A strong such selection process most likely exists in wild animals under tough environmental conditions but not in domestic animals and in humans.
I agree on the observation – allergy is found only in humans and domesticated animals – while the explanation is implausible as it cannot be generalized to humans. As vitamin D supplements are both used for humans, cats, dogs, horses and lab mice, it is a more likely explanation in particular as we have now have 4 randomized trials in humans confirming the hypothesis.
Der DAK Kinder und Jugendreport
Der neue DAK Gesundheitsreport zu Kindern und Jugendlichen ist etwas besonderes. Die meisten internationaler Surveys, ISAAC mal ausgenommen, umfassten weniger als 10.000 Probanden oder konzentriert sich auf bestimmte Altersgruppen. Die KiGGS-Studie als bekannte, große nationale Studie arbeitete im Wesentlichen mit Fragebögen, welche die Eltern und die Teilnehmer selbst ab elf Jahre ausfüllen konnten. Kleinere nationale Auswertungen auf Basis von GKV-Abrechnungsdaten gab es bisher in der Regel nur mit thematischem Fokus auf bestimmte Erkrankungsindikationen und ohne Längsschnitt. Neu, jedenfalls für deutsche Verhältnisse, ist die Analyse auf Basis der Abrechnungs- und Versorgungsdaten von rund 600.000 DAK- versicherten Kindern und 430.000 Eltern, zuerst hier im Querschnitt, später auch im Längsschnitt.
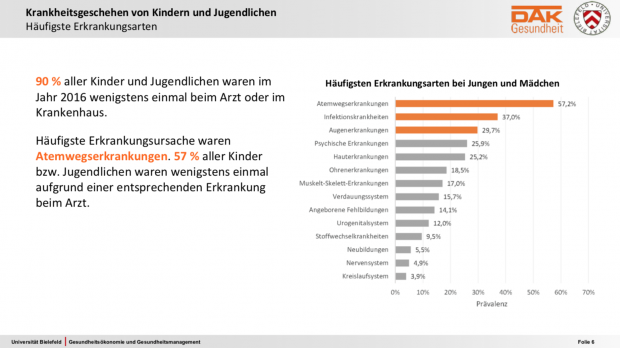
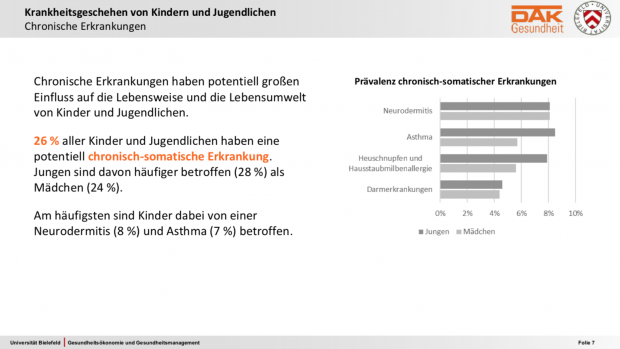

Interessant auch, dass hier mehr Kinder auf dem Land Heuschnupfen hatten – vielleicht sollte Arte mal die Agenda überarbeiten…
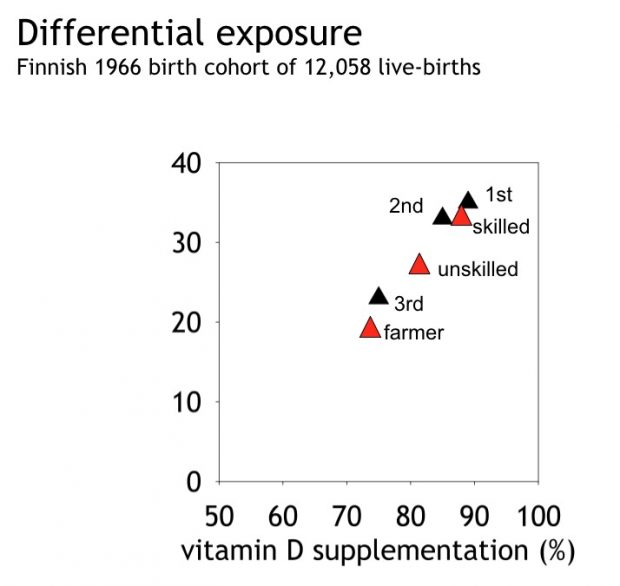
Vitamin D and farming
We published already in 2004 that farmers use less vitamin D supplements as they spend so much time outdoors
and give their babies more raw milk avoiding supplemented milk. Just for the notes.
Unseriöse Nestlé Werbung für HA Nahrung
Durch eine neuere BMJ Arbeit kam ich auf zufällig auf www.babyservice.d Laut Impressum ist der Dienstanbieter dieser Website Nestlé Nutrition GmbH, Lyoner Straße 23, D-60523 Frankfurt am Main.
Die Behauptung hier
Der positive Effekt der Säuglingsnahrung mit partiell aufgespaltenem Molkeneiweiß hält bis zum Alter von 15 Jahren an. Quelle: von Berg et al. (2015).
ist falsch wie ich im Sommer in PAI (paywall) vorgerechnet habe.
Using a more appropriate control group as demonstrated here, the GINI study can not claim any benefit of hydrolyzed formulas. Even worse, some children may experience negative side effects of hydrolyzed formulas as shown by several case reports.
Did we trade rickets with allergy?
I have written on that before but find it striking again when reading another historical perspective.
The first insight into the possible relationship between the industrialization of Northern Europe and rickets was made by Sniadecki in 1822 when he concluded that children who lived in the inner city of Warsaw had a high incidence of rickets because of their lack of sun exposure. This was based on his clinical observations that children living in rural areas outside of Warsaw did not suffer from rickets while children born and raised in Warsaw were plagued with the disease.
Now we are supplementing vitamin D to nearly all inner city children who have a high allergy prevalence but not so much in rural areas where we see less allergy – everywhere, not only in Warsaw.
Did we trade rickets with allergy?
A bad study is worse than no study
“Direct infant UV light exposure is associated with eczema and immune development”. Kristina Rueter, Anderson P. Jones, Aris Siafarikas, Ee-Mun Lim, Natasha Bear, Paul S. Noakes, Susan L. Prescott and Debra J. Palmer.
Article in press 2018 American Academy of Allergy, Asthma & Immunology https://doi.org/10.1016/j.jaci.2018.08.037
These graphical abstracts look a bit strange like “science for dummies”. Isn’t there a major discrepancy of title and abstract?
IMHO this is a RCT of vitamin D supplementation of newborns and not a study of UV light exposure. Maybe the authors needed a selling point for a poorly designed study?
The (only) allergy outcome is shown in table. 6 of 90 in the placebo group and 9 of 90 in the vitamin group develop eczema. This translates into an OR of 1.6 (0.5-4.6, P = 0.4214). I read this as a non significant association of exposure and outcome which is quite understandable given
1. the low power of the study. My result of a post hoc power calculation is around 12%.
2. the ignorance of the main eczema risk factor ( filaggrin mutation!). Allocation by a “history of maternal allergic disease” does not allocate filaggrin mutations equally between groups.
3. the ignorance of maternal vitamin D levels. Restricting to maternal levels >50 nmol/L introduced as a bias toward supplemented fetuses.
4. the ignorance of vitamin D fed by formula. So clearly this is more a dose-finding study and not a RCT of vitamin D supplementation as also the controls are (heavily) exposed.
5. the ignorance of the most relevant outcome in this age group (which is sensitization against food allergens).
6. the trial registration number is wrong.
7. the flowchart numbers have errors, for example the size of the vitamin D group at 3 months need to be N=91 and not N=90.
8. the “vitamin D hypothesis” did not emerge to explain associations found between regions of higher latitudes and increased risk of development of allergic diseases in children – the hypothesis emerged by theoretical considerations of the immune effects of artificial vitamin D supplementation.
JACI – how did that survive your review?
17.1.2020 Update
As it turned out, I didn’t find all issue, there are even more when reading now the review of Maslin et al.
Does Acinetobacter lwoffii F78 protect from allergy?
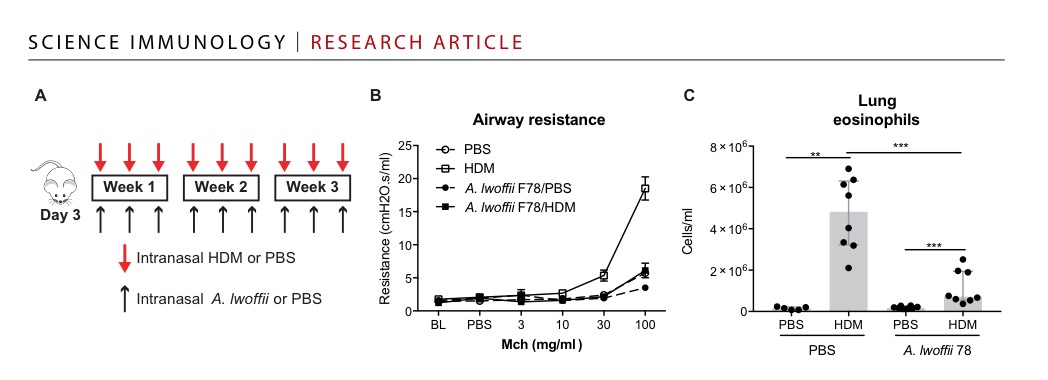
When reading a new Science immunology paper (“Inception of early-life allergen-induced airway hyperresponsiveness is reliant on IL-13+CD4+T cells“) one could again think that A. lwoffii could protect from the development of house mite allergy.
The paper, however, leaves it open (even doesn’t mention the result in the discussion) if this is any specific A. lwofii effect or just some some LPS effect that antagonized the vitamin D containing food.
So no news even 10 years after the initial Acinetobacter hype. The only verified fact remain several deaths caused by Acinetobacter in newborns.
Finding the allergy cause
Genomics did not really help to explain allergic mechanisms beyond IL33. But combining now stem cell & immune cell Identity tracking looks like a promising strategy for identifying initial disease events. At least colleagues at the MDC think so.
LifeTime – ein visionärer Vorschlag für ein EU-Flagschiff. Zuverlässig vorherzusagen, wann eine Krankheit ausbricht oder wie sie verläuft, erscheint wie ein Traum. Ein europäisches Konsortium will ihn Wirklichkeit werden lassen und dabei vor allem neue Technologien der Einzelzellbiologie nutzen. Führende Forscherinnen und Forscher haben daher einen Antrag für ein FET-Flagschiff mit dem Namen LifeTime eingereicht.