The Guardian writes that humans age dramatically increases in two bursts at 44, then 60 years. The headline may be misleading as it misses the growth spurt in 10 year old children but also the cachexia of people around 90.
The report relies on a report with limited observation period
In this study, we performed comprehensive multi-omics profiling on a longitudinal human cohort of 108 participants, aged between 25 years and 75 years. The participants resided in California, United States, and were tracked for a median period of 1.7 years, with a maximum follow-up duration of 6.8 years.
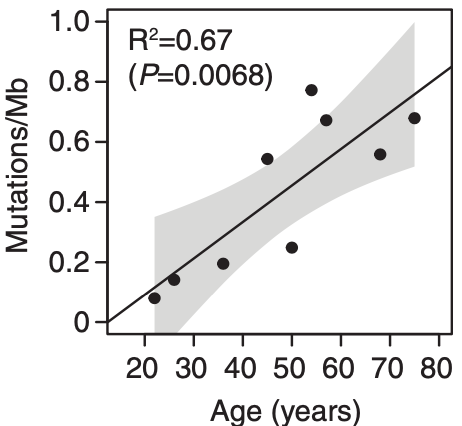
but nevertheless it confirms my own observation with age effects in bursts around 45 and again in 60 (even did some studies on chronobiology and DNA ageing that confirmed ELOVL2 effects in 2015 while a confirmation study 2020 could not be finished due to COVID19).
It is a bit annoying, however, as there are always different numbers in the Snyder study when compared to earlier reports of the same group that missed the current findings.
Even worse as there was no a priori study plan the results look more like a display of a random data warehouse where you can pick what you waant. So where is the main effect and what is the driving force? The authors are counting significant changes

If we look at the proportional max/min change it is roughly 40% at transcriptomics and proteomics and 32% at metabolomics level. But I have no clue what’s behind these peaks – GTPase activity? Oxygen carrier? As there are no methylation data, no tissue biospy, not even any blood sample covering the whole time span, it remains a rather fuzzy paper that tries to provide a scientific basis of a common sense observation.